A) beta emission
B) gamma emission
C) positron emission
D) electron capture
E) Any of the above processes will lower the neutron-to-proton ratio.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In what type of radioactive decay does the atomic number of the product increase by one?
A) alpha
B) beta
C) gamma
D) positron emission
E) electron capture
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Due to the nature of the positron,________ is actually detected in positron emission tomography.
A) alpha radiation
B) beta radiation
C) gamma radiation
D) x-ray emission
E) neutron emission
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bombardment of uranium-238 with a deuteron (hydrogen-2) generates neptunium-237 and ________ neutrons.
A) 1
B) 2
C) 3
D) 4
E) 5
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the nuclear transmutation represented by
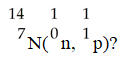 ,what is the emitted particle?
,what is the emitted particle?
A) neutron
B) proton
C) positron
D) alpha particle
E) electron
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is emitted in the nuclear transmutation,
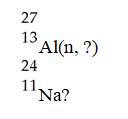
A) an alpha particle
B) a beta particle
C) a neutron
D) a proton
E) a gamma photon
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half-life of cobalt-60 is 5.20 yr.How many milligrams of a 2.000-mg sample remain after 9.50 years?
A) 0.565
B) 7.03 × 10-22
C) 7.076
D) 1.095
E) 1.435
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The missing product in this reaction would be found in which group of the periodic table? 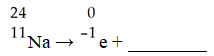
A) 1A
B) 2A
C) 3A
D) 8A
E) 7A
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms with the same atomic number and different mass numbers ________.
A) do not exist
B) are isomers
C) are isotopes
D) are allotropes
E) are resonance structures
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alpha decay produces a new nucleus whose ________ than those respectively of the original nucleus.
A) atomic number is 2 less and mass number is 2 less
B) atomic number is 1 less and mass number is 2 less
C) atomic number is 2 less and mass number is 4 less
D) atomic number is 2 more and mass number is 4 more
E) atomic number is 2 more and mass number is 2 less
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Radium undergoes alpha decay.The product of this reaction also undergoes alpha decay.What is the product of this second decay reaction?
A) Po
B) Rn
C) U
D) Th
E) Hg
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What exposure level to radiation is fatal to most humans?
A) 100 rem
B) 200 rem
C) 600 rem
D) 300 rem
E) 1000 rem
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
The initial element used to make cobalt-60 for cancer radiation therapy is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
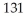 I has a half-life of 8.04 days.Assuming you start with a 1.03 mg sample of
I has a half-life of 8.04 days.Assuming you start with a 1.03 mg sample of  I,how many mg will remain after 13.0 days?
I,how many mg will remain after 13.0 days?
A) 0.326
B) 0.268
C) 0.422
D) 0.0781
E) 0.336
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following devices converts radioactive emissions to light for detection?
A) Geiger counter
B) photographic film
C) scintillation counter
D) radiotracer
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All atoms of a given element have the same ________.
A) mass number
B) number of nucleons
C) atomic mass
D) number of neutrons
E) atomic number
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
What happens in the nucleus of an atom that undergoes positron emission?
Correct Answer

verified
A proton is converte...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of these nuclides is most likely to be radioactive?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 161 - 178 of 178
Related Exams