A) 6.78
B) 2.76
C) 1.60
D) 0.192
E) 4.02
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A fixed amount of gas at 25.0 °C occupies a volume of 10.0 L when the pressure is 751 torr.Use Boyle's law to calculate the pressure (torr) when the volume is reduced to 7.25 L at a constant temperature of 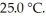
A) 1.04 × 103
B) 0.097
C) 5.44 × ![]()
D) 544
E) 1.36
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 50.75 g of a gas occupies 10.0 L at STP,129.3 g of the gas will occupy ________ L at STP.
A) 3.92
B) 50.8
C) 12.9
D) 25.5
E) 5.08
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a unit of pressure?
A) mm Hg
B) mm
C) atm
D) Pa
E) psi
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The average kinetic energy of the particles of a gas is ________ proportional to ________.
A) directly, the rms speed
B) directly, the square root of the rms speed
C) inversely, the square of the rms speed
D) directly, the square of the rms speed
E) inversely, the particle mass
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Two deviations of real gases from ideal gases which are treated in the van der Waals equation are finite molecular volume and non-zero molecular attractions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas mixture of N2 and H2 has a total pressure of 9.40 atm and contains 11.3 mol of gas.If the partial pressure of N2 is 4.89 atm,how many moles of H2 are in the mixture?
A) 4.51
B) 5.65
C) 5.42
D) 11.3
E) 5.88
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following gases in order of increasing average molecular speed at 25 °C. He,O2,CO2,N2
A) He < N2 < O2 < CO2
B) He < O2 < N2 < CO2
C) CO2 < O2 < N2 < He
D) CO2 < N2 < O2 < He
E) CO2 < He < N2 < O2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas originally at 27 °C and 1.00 atm pressure in a 3.3 L flask is cooled at constant pressure until the temperature is 11 °C.The new volume of the gas is ________ L.
A) 0.27
B) 3.1
C) 3.3
D) 3.5
E) 0.24
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A flask contains a mixture of N2 and H2 at a total pressure of 5.20 atm.If there are 1.00 mol of H2 and 8.00 mol of N2 in the flask,what is the partial pressure (atm) of N2?
A) 2.60
B) 4.62
C) 5.20
D) 8.00
E) 0.578
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
760 torr is equivalent to ________ mm Hg.
A) 380
B) 4.78 × 103
C) 1
D) 760
E) 121
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A balloon originally had a volume of 4.39 L at 44 °C and a pressure of 729 torr.The balloon must be cooled to ________ °C to reduce its volume to 3.99 L (at constant pressure) .
A) 0.00
B) 15.1
C) -288
D) 288
E) 546
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following equations shows an incorrect relationship between pressures given in terms of different units?
A) 1.20 atm = 122 kPa
B) 152 mm Hg = 2.03 × 104 Pa
C) 0.760 atm = 578 mm Hg
D) 1.0 torr = 2.00 mm Hg
E) 1.00 atm = 760 torr
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following gases would deviate the least from ideal gas behavior?
A) Ne
B) CH3Cl
C) Kr
D) CO2
E) F2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The main component of air is oxygen.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the rms speed (m/s)of oxygen molecules at 36.0 °C?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement about atmospheric pressure is false?
A) As air becomes thinner, its density decreases.
B) Air actually has weight.
C) With an increase in altitude, atmospheric pressure increases as well.
D) The warmer the air, the lower the atmospheric pressure.
E) Atmospheric pressure prevents water in lakes, rivers, and oceans from boiling away.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume (mL) of sulfur dioxide can be produced by the complete reaction of 3.82 g of calcium sulfite with excess HCl (aq) ,when the final SO2 pressure is 827 torr at 44.0 °C?
A) 7.60 × 102
B) 1.39 × 10-4
C) 1.00 × 10-3
D) 0.106
E) 5.78 × 102
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If pressure and temperature are kept constant,the reaction of 38 mL of Cl2 gas with 22 mL of CH4 gas via the equation: Cl2 (g) + CH4 (g) → HCl (g) + CH3Cl (g) Will produce a total of ________ mL of products.
A) 16
B) 30
C) 60
D) 38
E) 22
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of CO2 gas (3.0 mol) effused through a pinhole in 18.0 s.It will take ________ s for the same amount of H2 to effuse under the same conditions.
A) 396
B) 3.84
C) 0.818
D) 84.4
E) 1.92
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 175
Related Exams