A) If we know that a reaction is an elementary reaction, then we know its rate law.
B) The rate-determining step of a reaction is the rate of the fastest elementary step of its mechanism.
C) Since intermediate compounds can be formed, the chemical equations for the elementary reactions in a multistep mechanism do not always have to add to give the chemical equation of the overall process.
D) In a reaction mechanism, an intermediate is identical to an activated complex.
E) All of the above statements are true.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A catalyst that is present in the same phase as the reacting molecules is called a __________ catalyst.
Correct Answer

verified
homogeneous
Correct Answer
verified
Multiple Choice
The average rate of disappearance of I- between 1200.0 s and 1600.0 s is __________ M/s.
A) 1.8 × 10-5
B) 1.2 × 10-5
C) 2.0 × 10-5
D) 5.0 × 104
E) 1.6 × 10-4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isomerization of methylisonitrile to acetonitrile CH3NC (g) → CH3CN (g) Is first order in CH3NC. The rate constant for the reaction is 9.45 × 10-5s-1 at 478 K. The half-life of the reaction when the initial [CH3NC] is 0.030 M is __________ s.
A) 1.06 × 104
B) 5.29 × 103
C) 3.53E × 105
D) 7.33 × 103
E) 1.36 × 10-4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate constant for a particular second-order reaction is 0.47M-1s-1. If the initial concentration of reactant is 0.25 mol/L, it takes __________ s for the concentration to decrease to 0.080 mol/L.
A) 18
B) 1.4
C) 8.5
D) 4.0
E) 0.08
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrogen dioxide decomposes to nitric oxide and oxygen via the reaction: 2NO2 → 2NO + O2 In a particular experiment at 300 °C, [NO2] drops from 0.0100 to 0.00650 M in 100 s. The rate of disappearance of NO2 for this period is __________ M/s.
A) 0.35
B) 3.5 × 10-3
C) 3.5 × 10-5
D) 7.0 × 10-3
E) 1.8 × 10-3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The enzyme nitrogenase converts __________ into __________.
A) ammonia, urea
B) CO and unburned hydrocarbons, H2O and CO2
C) nitrogen, ammonia
D) nitrogen oxides, N2 and O2
E) nitroglycerine, nitric acid, and glycerine
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate of disappearance of HBr in the gas phase reaction 2HBr (g) → H2 (g) + Br2 (g) Is 0.130 Ms-1 at 150°C. The rate of reaction is __________ Ms-1.
A) 3.85
B) 0.0650
C) 0.0169
D) 0.260
E) 0.0860
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half-life of this reaction is __________ s.
A) 0.97
B) 7.1
C) 5.0
D) 3.0
E) 0.14
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A possible mechanism for the overall reaction Br2 (g) + 2NO (g) → 2NOBr (g)
Is
NO (g) + Br2 (g) ![A possible mechanism for the overall reaction Br<sub>2</sub> (g) + 2NO (g) → 2NOBr (g) Is NO (g) + Br<sub>2</sub> (g) NOBr<sub>2</sub> (g) (fast) NOBr<sub>2</sub> (g) + NO (g) 2NOBr (slow) The rate law for formation of NOBr based on this mechanism is rate = __________. A) k<sub>1</sub>[NO]1/2 B) k<sub>1</sub>[Br<sub>2</sub>]1/2 C) (k<sub>2</sub>k<sub>1</sub>/k<sup>-1</sup>) [NO]<sup>2</sup>[Br<sub>2</sub>] D) (k<sub>1</sub>/k<sup>-1</sup>) <sup>2</sup>[NO]<sup>2</sup> E) (k<sub>2</sub>k<sub>1</sub>/k<sup>-1</sup>) [NO]<sup>2</sup>[Br<sub>2</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB1822/11ea7ce9_fb1b_ab4c_b46a_17c0e2000001_TB1822_11.jpg) NOBr2 (g) (fast)
NOBr2 (g) + NO (g)
NOBr2 (g) (fast)
NOBr2 (g) + NO (g) ![A possible mechanism for the overall reaction Br<sub>2</sub> (g) + 2NO (g) → 2NOBr (g) Is NO (g) + Br<sub>2</sub> (g) NOBr<sub>2</sub> (g) (fast) NOBr<sub>2</sub> (g) + NO (g) 2NOBr (slow) The rate law for formation of NOBr based on this mechanism is rate = __________. A) k<sub>1</sub>[NO]1/2 B) k<sub>1</sub>[Br<sub>2</sub>]1/2 C) (k<sub>2</sub>k<sub>1</sub>/k<sup>-1</sup>) [NO]<sup>2</sup>[Br<sub>2</sub>] D) (k<sub>1</sub>/k<sup>-1</sup>) <sup>2</sup>[NO]<sup>2</sup> E) (k<sub>2</sub>k<sub>1</sub>/k<sup>-1</sup>) [NO]<sup>2</sup>[Br<sub>2</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB1822/11ea7ce9_fb1b_d25d_b46a_a95f75214c0e_TB1822_11.jpg) 2NOBr (slow)
The rate law for formation of NOBr based on this mechanism is rate = __________.
2NOBr (slow)
The rate law for formation of NOBr based on this mechanism is rate = __________.
A) k1[NO]1/2
B) k1[Br2]1/2
C) (k2k1/k-1) [NO]2[Br2]
D) (k1/k-1) 2[NO]2
E) (k2k1/k-1) [NO]2[Br2]2
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the magnitude of the rate constant for the reaction?
A) 1.15 × 104
B) 4.6
C) 230
D) 115
E) 713
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substance in the reaction below either appears or disappears the fastest? 4NH3 + 7O2 → 4NO2 + 6H2O
A) NH3
B) O2
C) NO2
D) H2O
E) The rates of appearance/disappearance are the same for all of these.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following reaction: A → 2C The average rate of appearance of C is given by Δ[C]/Δt. Comparing the rate of appearance of C and the rate of disappearance of A, we get Δ[C]/Δt = __________ × (-Δ[A]/Δt) .
A) +2
B) -1
C) +1
D) +1/2
E) -1/2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction below is first order in [H2O2]: 2H2O2 (l) → 2H2O (l) + O2 (g) A solution originally at 0.600 M H2O2 is found to be 0.075 M after 54 min. The half-life for this reaction is __________ min.
A) 6.8
B) 18
C) 14
D) 28
E) 54
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The elementary reaction 2NO2 (g) → 2NO (g) + O2 (g) Is second order in NO2 and the rate constant at 501 K is 7.93 × 10-3M-1s-1. The reaction half-life at this temperature when [NO2]0= 0.45 M is __________ s.
A) 3.6 × 10-3
B) 0.011
C) 126
D) 87
E) 280
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The average rate disappearance of A between 20 s and 30 s is __________ mol/s.
A) 5.0 × 10-4
B) 1.6 × 10-2
C) 1.5 × 10-3
D) 670
E) 0.15
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate constant of a first-order process that has a half-life of 3.50 min is __________ s-1.
A) 0.693
B) 1.65 × 10-2
C) 1.98
D) .198
E) 3.30 × 10-3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At elevated temperatures, methylisonitrile (CH3NC) isomerizes to acetonitrile (CH3CN) : CH3NC (g) → CH3CN (g)
The reaction is first order in methylisonitrile. The attached graph shows data for the reaction obtained at 198.9°C. 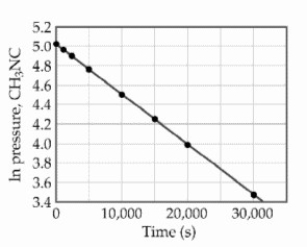 The rate constant for the reaction is __________ s-1.
The rate constant for the reaction is __________ s-1.
A) -1.9 × 104
B) +1.9 × 104
C) -5.2 × 10-5
D) +5.2 × 10-5
E) +6.2
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Short Answer
If a rate law is second order (reactant), doubling the reactant __________ the reaction rate.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Under constant conditions, the half-life of a first-order reaction __________.
A) is the time necessary for the reactant concentration to drop to half its original value
B) is constant
C) can be calculated from the reaction rate constant
D) does not depend on the initial reactant concentration
E) All of the above are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 130
Related Exams