Correct Answer

verified
Correct Answer
verified
Essay
Write a Lewis structure for the chlorite ion, ClO2-, that obeys the octet rule, showing all non-zero formal charges, and give the total number of resonance structures for ClO2- that obey the octet rule.
Correct Answer

verified
 Total number of res...
Total number of res...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Of the species NO2, NO, and N2, only NO2 is an exception to the octet rule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming the octet rule is obeyed, how many covalent bonds will a carbon atom form to give a formal charge of zero?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be a covalent compound?
A) KF
B) CaCl2
C) SF4
D) Al2O3
E) CaSO4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond in which of the following pairs of atoms would have the greatest percent ionic character (i.e., most polar) ?
A) C - O
B) S - O
C) Na - I
D) Na - Br
E) F - F
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the Lewis structure of the iodate ion, IO3-, that satisfies the octet rule, the formal charge on the central iodine atom is
A) +2
B) +1
C) 0
D) -1
E) -2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the elements listed below has the greatest electronegativity?
A) Mg
B) Ga
C) Si
D) Ba
E) Pb
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the chloride ion is
A) ![]()
B) ![]() -
-
C) ![]() -
-
D) ![]() -
-
E) Cl-
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Of the following substances, KCl, KBr, and KF, KF will have the highest melting point.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances will display an incomplete octet in its Lewis structure?
A) CO2
B) Cl2
C) ICl
D) NO
E) SO2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many covalent bonds will be drawn to phosphorous in PO43- for the dot structure that expands the octet to minimize formal charge and if necessary places negative formal charges on the most electronegative atom(s) .
A) 4
B) 5
C) 6
D) 7
E) 8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound?
A) ClF3
B) FeCl3
C) NH3
D) PF3
E) SO3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Write the Lewis structure of ammonia (nitrogen trihydride).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has a Lewis structure that does not obey the octet rule?
A) N2O
B) CS2
C) PH3
D) CCl4
E) NO2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has an atom with an incomplete octet?
A) NF3
B) H2O
C) AsCl3
D) GeH4
E) BF3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
BeF42- is called the fluoberyllate ion. The formal charge on the beryllium atom in this ion is
A) -2
B) -1
C) 0
D) +1
E) +2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Consider the hypothetical element X with electron-dot formula 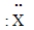 Give the formula for the simplest compound this element forms with chlorine.
Give the formula for the simplest compound this element forms with chlorine.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which response includes all the molecules below that do not follow the octet rule? (1) H2S (2) BCl3 (3) PH3 (4) SF4
A) (2) and (4)
B) (2) and (3)
C) (1) and (2)
D) (3) and (4)
E) (1) and (4)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of resonance structures for the sulfur dioxide molecule that satisfy the octet rule is
A) 1
B) 2
C) 3
D) 4
E) None of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 118
Related Exams