A) protons are transferred from one atom to another.
B) neutrons are shared between two atoms to form an isotope.
C) electrons are shared between two atoms to complete their octets.
D) the hydrogen of one water molecule is attracted to the oxygen of another water molecule.
E) electrons are transferred from one atom to another.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A coke has a pH of 3.5.This means that it has an excess of _______ ions and would be called a(n) _____.
A) H+; acid
B) OH-; acid
C) H+; base
D) OH-; base
E) H+; neutral solution
G) A) and E)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Sulfur has an atomic number of 16.What would be the valence number of this element?1_01_2012
A) One
B) Two
C) Three
D) Four
E) Six
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these does not occur when a sodium atom transfers an electron to a chlorine atom?
A) The sodium atom becomes a positively charged ion.
B) The positive and negative ions will attract each other, forming a crystal if no water is present.
C) The ions will separate in the presence of water.
D) There is a sharing of the electrons between the sodium and chlorine atoms.
E) The chlorine atom becomes a negatively charged ion.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You are caring for the freshwater aquarium of a friend while they are on vacation.You are told to check the pH of the tank and make sure that it stays between 6.5 and 7.0.In order to ensure that you do not have to worry about the pH you
A) treat the fish and the tank with aspirin.
B) add NaOH so that changes in the tank are calculated.
C) add HCl so that you can track the changes in the tank's pH.
D) do nothing since the pH will not change unless you do something wrong and you plan to follow the directions exactly.
E) add a buffer so changes in pH are minimized.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The type of bond that would form from the transfer of an electron from one atom to another,as depicted in the figure,is a 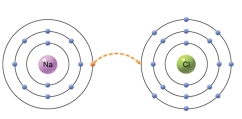
A) covalent.
B) ionic.
C) hydrogen.
D) atomic.
E) isotope.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The term to describe any substance,which can prevent the pH of a solution from changing by either releasing or absorbing H+ in a solution is
A) equalizer.
B) solute.
C) buffer.
D) acid.
E) base.
G) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following is not a contributing event in the formation of acid rain?
A) Fossil fuels consume oxygen removing it from the atmosphere.
B) Coal & oil emit sulfur dioxide and nitrogen oxide when they are burned.
C) Fossil fuel emission gases combine with water in the atmosphere.
D) A solution of sulfuric acid and nitric acid forms.
E) Precipitation containing sulfuric acid and nitric acid fall to the Earth.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The figure below is depicting the interaction of water molecules with one another,which involves the use of 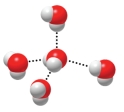
A) covalent bonds.
B) hydrogen bonds.
C) ionic bonds.
D) valence shells.
E) solvents.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isotopes of an element differ in their
A) proton number.
B) electron number.
C) neutron number.
D) type of bonds.
E) atomic number.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom with a neutral charge has
A) equal numbers of neutrons and electrons.
B) more neutrons making it more neutral.
C) the same number of protons and neutrons.
D) equal numbers of protons and electrons.
E) more protons than it does electrons.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Regarding atoms,identify which statement below is correct.
A) An element may be composed of several types of atoms.
B) The nucleus of an atom contains protons and electrons.
C) The number and arrangement of electrons in an atom governs its chemical activity.
D) The positive charges of an element are carried by the electrons.
E) The neutral charges of an element are carried by the protons.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Engineers design city sidewalks using blocks of concrete separated by a small gap to prevent them from cracking.What property of water is being taken into account for this design? Water
A) is a good solvent.
B) is less dense when it is frozen.
C) is cohesive.
D) is resistant to temperature changes.
E) has a high heat capacity.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not one of the most common elements in living things?
A) carbon
B) oxygen
C) hydrogen
D) iron
E) nitrogen Six elements of matter make up the majority of body weight of most organisms. Their presence and properties are essential to the uniqueness of living things.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements differ from each other in their
A) physical properties only.
B) atomic number only.
C) type of subatomic particles.
D) physical properties and atomic number.
E) type of electrons.
G) A) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which one of the following is the smallest unit of matter that has all the properties of an element?
A) molecule
B) element
C) atom
D) compound
E) electron
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In what ways are radioactive isotopes potentially harmful?
A) Unmonitored release into the environment can make changes in a cell's DNA.
B) They are used to trace molecular changes.
C) They are used to destroy abnormal cells.
D) They are used to determine the age of biological specimens.
E) They are used to trace the path of materials throughout the body.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What property of water causes sugar to dissolve in coffee?
A) Water has high heat capacity.
B) Water is less dense as ice.
C) Water is a good solvent.
D) Water is cohesive.
E) Water is able to change states.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do cells need buffering agents?
A) to minimize the changes in pH of their internal environment
B) to operate at a constant pH of 2.0
C) to carry out life functions in extremely acidic conditions
D) to help transfer electrons from one atom to another
E) to increase the amount of OH- in their surroundings
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chemical bonds involve
A) the giving and taking of electrons.
B) the giving and taking of protons.
C) the giving, taking or sharing of electrons.
D) the giving, taking or sharing of protons.
E) the sharing of electrons.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 64
Related Exams