A) C2H5OH(l)
B) C2H2(g)
C) C3H8(g)
D) C3H7OH(l)
E) C2H6(g)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a chemical reaction to be spontaneous only at high temperatures, which conditions must be met?
A) ΔS° > 0, ΔH° > 0
B) ΔS° > 0, ΔH° < 0
C) ΔS° < 0, ΔH° < 0
D) ΔS° < 0, ΔH° > 0
E) ΔG° > 0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG°rxn at 298 K for the following reaction? 2Cl2(g) + SO2(g) → SOCl2(g) + Cl2O(g) 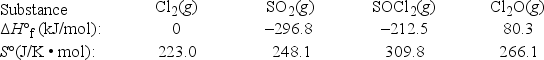
A) 129.3 kJ/mol
B) 133.4 kJ/mol
C) 196.0 kJ/mol
D) 199.8 kJ/mol
E) 229.6 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat of vaporization of 1-pentanol is 55.5 kJ/mol, and its entropy of vaporization is 148 J/K•mol. What is the approximate boiling point of 1-pentanol?
A) 100oC
B) 375 oC
C) 0 oC
D) 25 oC
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
HI has a normal boiling point of -35.4°C, and its ΔHvap is 21.16 kJ/mol. What is the molar entropy of vaporization (ΔSvap) of HI?
A) 598 J/K • mol
B) 68.6 J/K • mol
C) 75.2 J/K • mol
D) 0.068 J/K • mol
E) 89.0 J/K • mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The reaction SiO2(s) + Pb(s) → PbO2(s) + Si(s) is spontaneous: ΔG°f (PbO2(s)) = -217 kJ/mol ΔG°f (SiO2(s)) = -856 kJ/mol
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure below which shows ΔG° for a chemical process plotted against absolute temperature. Which one of the following is an incorrect conclusion, based on the information in the diagram? 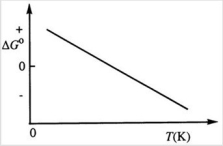
A) ΔH° > 0.
B) ΔS° > 0.
C) The reaction is spontaneous at high temperatures.
D) ΔS° increases with temperature while ΔH° remains constant.
E) There exists a certain temperature at which ΔH° = TΔS°.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
At equilibrium, ΔG° = 0.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium carbonate can be made by heating sodium bicarbonate: 2NaHCO3(s) → Na2CO3(s) + CO2(g) + H2O(g) At 25°C, for this reaction, ΔH°rxn = 128.9 kJ/mol and ΔG°rxn = 33.1 kJ/mol. Above what minimum temperature will the reaction become spontaneous under standard state conditions?
A) 0.4 K
B) 3.9 K
C) 321 K
D) 401 K
E) 525 K
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ozone (O3) in the atmosphere can react with nitric oxide (NO) as follows. O3(g) + NO(g) → NO2(g) + O2(g) . ΔH°rxn = -199 kJ/mol, ΔS°rxn = -4.1 J/K • mol What is ΔG°rxn for this reaction at 25°C?
A) 1020 kJ/mol
B) -1.22 × 103 kJ/mol
C) 2.00 × 103 kJ/mol
D) -1.42 × 103 kJ/mol
E) -198 kJ/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an example of a nonspontaneous process?
A) Ice melting at room temperature
B) Sodium metal reacting violently with water
C) Rusting of iron at room temperature
D) A ball rolling downhill
E) Water freezing at room temperature
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
How does the entropy change when a gas is liquified?
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the pressure required for standard entropy conditions.
Correct Answer

verified
Correct Answer
verified
True/False
The higher the pressure of a gas sample, the greater is its entropy.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The following reaction is spontaneous under standard state conditions at 25°C: AgCl(s) → Ag+ (aq) + Cl- (aq) (ΔG° = 55 kJ/mol)
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has ΔG°f = 0 at 25oC?
A) H2O(l)
B) H2O(g)
C) Na(s)
D) O3(g)
E) O(g)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is defined as a measure of the randomness or disorder of a system?
A) Gibbs free energy
B) Entropy
C) Spontaneous process
D) Nonspontaneous process
E) Enthalpy
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG°rxn for the following reaction? 2NO(g) + Cl2(g) → 2NOCl(g) 
A) -40.40 kJ/mol
B) +40.40 kJ/mol
C) -20.20 kJ/mol
D) +20.20 kJ/mol
E) +152.90 kJ/mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The absolute standard entropy of atom X(g) is 100 J/K • mol. Which is a reasonable value for the absolute standard entropy of X2(g) ?
A) 200 J/K • mol
B) 170 J/K • mol
C) 100 J/K • mol
D) 80 J/K • mol
E) 50 J/K • mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The entropy change ΔS° at 298 K for the reaction NH4Cl(s) →NH3(g) + HCl(g) is negative.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 107
Related Exams