A) Number of protons
B) Number of neutrons
C) They are isotopes.
D) Number of electrons
E) They are all ions.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium reacts vigorously with water to form hydrogen gas and a compound containing sodium ions.Which other element is expected to react with water in a similar way?
A) hydrogen
B) aluminum
C) nitrogen
D) potassium
E) silicon
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The overall charge of an atom is ________ if the number of electrons is ________ than the number of protons.
A) negative, less
B) positive, greater
C) positive, less
D) neutral, less
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements does not apply to metalloids?
A) The physical properties of metalloids resemble those of a metal.
B) All metalloids are electrical insulators.
C) Metalloids lie along the stair-step line beginning at boron.
D) The chemical properties of metalloids are similar to nonmetals.
E) Metalloids are also known as semi-metals.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One balloon is filled with CO2, while the other contains H2.They are filled to equal volumes and contain the same number of molecules.Predict the relative densities of the gases in the two balloons.
A) The CO2 gas should be 44 times the density of the H2 gas.
B) The CO2 gas should be 22 times the density of the H2 gas.
C) The CO2 gas should be 1.5 times the density of the H2 gas.
D) The H2 gas should be 0.67 times the density of the CO2 gas.
E) The gases should have the same density.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about Mendeleev's periodic table is incorrect?
A) Mendeleev arranged the known elements in order of increasing relative atomic mass.
B) He grouped elements with similar properties into columns and rows so that their properties varied in a regular pattern.
C) He arranged the elements so that they were in increasing atomic number order.
D) He was able to predict the existence and properties of several elements that were unknown at the time.
E) Mendeleev developed his table before the discovery of protons.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford's scattering experiment demonstrated
A) the existence of protons.
B) the existence of electrons.
C) the existence of neutrons.
D) that most of the mass of an atom is in its nucleus.
E) that the charge-to-mass ratio of an electron is constant.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements regarding the nucleus of the atom is incorrect?
A) The nucleus is the central core of the atom.
B) The nucleus contains the electrons and the protons.
C) The nucleus contains most of the mass of the atom.
D) The nucleus contains the neutrons.
E) The nucleus contains the neutrons and protons and most of the mass of the atom.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The mass of exactly 100 carbon atoms is 12.01 amu.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The subatomic particles that make up the atom (of interest to chemists) include all of the following except the:
A) proton.
B) alpha particle.
C) electron.
D) neutron.
E) alpha particle and neutron.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
On the planet Melmac, in a galaxy far, far away, argon has three naturally occurring isotopes as follows: Isotope Mass (amu) Natural Abundance (%) Argon-40 39.962 74.20 Argon-38 37.963 15.15 Argon-36 35.968 10.65 What is the relative atomic mass of argon on Melmac?
A) 39.23 amu
B) 39.96 amu
C) 37.96 amu
D) 35.97 amu
E) 40.00 amu
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To the correct number of significant figures, the mass of exactly 250 atoms of mercury would be:
A) 200.6 amu
B) 250 amu
C) 5.015 x 104 amu
D) 5.0100 x 104 amu
E) 1.246 amu
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Boron has two isotopes: B-10 and B-11, with masses of 10.013 amu and 11.009 amu, respectively.The relative atomic mass of boron is 10.81 amu.Which statement best describes the percent abundance of the isotopes of boron?
A) It contains more B-10 than B-11.
B) It contains more B-11 than B-10.
C) It contains equal amounts of B-10 and B-11.
D) There must be a third isotope of boron.
E) A mass spectrum of boron is necessary to answer this question.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
This figure shows a chemical reaction taking place. 
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element that is an alkali metal in Period 3.
A) Na
B) Mg
C) Al
D) K
E) Ca
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements regarding ion formation is incorrect?
A) Nonmetals usually gain electrons to form ions that have a noble gas electron count.
B) Main-group metals usually lose electrons to form ions that have a noble gas electron count.
C) Elements in the same group often form ions of the same charge.
D) The charge of any element's ion can be simply predicted using the periodic table.
E) All of these statements are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The correct isotope symbol for the isotope in the figure is: 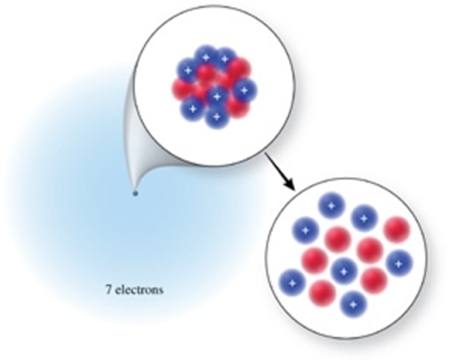
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms of different isotopes of a given element have the same
A) number of electrons.
B) sum of the number of protons and neutrons.
C) sum of the number of electrons and neutrons.
D) sum of the number of electrons, protons, and neutrons.
E) mass numbers.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To which class does the element chromium belong?
A) representative (main-group) elements
B) transition elements
C) lanthanides
D) actinides
E) metalloids
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ions of most main-group elements have the same number of ________ as the noble gas that is closest to them in the periodic table.
A) neutrons
B) protons
C) electrons
D) protons and electrons
E) neutrons and electrons
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 135
Related Exams